Sanofi (SNY), GSK Receive EU Approval for COVID-19 Vaccine
Sanofi SNY announced that the European Commission (“EC”), on Nov 10, approved VidPrevtyn Beta, its next-generation protein-based adjuvanted vaccine. The vaccine can be given as a booster for preventing COVID-19 in adults aged 18 years and older. The vaccine has been developed in partnership with GSK GSK.
Following the EC approval, VidPrevtyn Beta can be used as a booster dose in adults who have already been vaccinated with an mRNA-based vaccine developed by Pfizer PFE/BioNTech BNTX and Moderna or an adenoviral vaccine developed by AstraZeneca. Per Sanofi/GSK, the vaccine doses are ready to be shipped to member states of the EU.
The EC decision follows the positive opinion issued by the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) which also recommended approving the vaccine. The EC decision came a couple of hours after the CHMP recommended the vaccine. The EC and CHMP decisions are based on data from two immunobridging studies, which compared the immune response generated by the Sanofi/GSK partnered vaccine against authorized mRNA-based or adenoviral vaccines.
Based on its analysis of data, the CHMP and EC concluded that a booster dose of VidPrevtyn Beta is expected to be at least as effective as Pfizer/BioNTech’s Comirnaty at restoring protection against COVID-19.
Shares of Sanofi have lost 11.6% this year against the industry’s 3.7% rise.
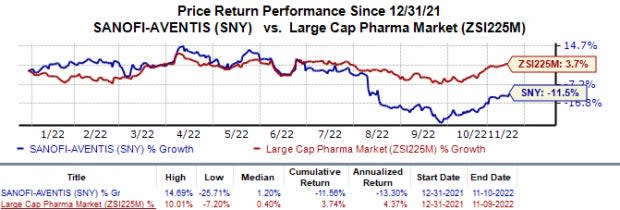
Image Source: Zacks Investment Research
VidPrevtyn Beta has been developed by Sanofi using its recombinant-protein technology platform and modeled on the Beta variant. The vaccine also utilizes GSK’s pandemic adjuvant platform.
We note that the approval comes when the COVID-19 infections are waning. In fact, the COVID-19 vaccine market is dominated by Pfizer/BioNTech and Moderna, which already have monovalent and bivalent vaccines authorized for use in the European Union. Pfizer/BioNTech and Moderna have already immunized millions of individuals across Europe and its BA.1 and BA.4/5 bivalent vaccines, which the EC has recently authorized, are in full force for supply to EU member states.
Sanofi Price
Sanofi price | Sanofi Quote
Zacks Rank
Sanofi currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Sanofi (SNY) : Free Stock Analysis Report
GSK PLC Sponsored ADR (GSK) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
BioNTech SE Sponsored ADR (BNTX) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

