Bristol Myers' (BMY) Onureg Gets European Commission Approval
Bristol Myers Squibb BMY announced that the European Commission (EC) has granted full Marketing Authorization to Onureg (azacitidine tablets).
Onureg is authorized as a maintenance therapy in adult patients with acute myeloid leukemia (AML) who achieved complete remission (CR) or complete remission with incomplete blood count recovery (CRi) following induction therapy with or without consolidation treatment and who are not candidates for, including those who choose not to proceed, hematopoietic stem cell transplantation (HSCT).
The approval was based on positive results from the QUAZAR AML-001 study, a phase III, international, randomized, double-blind study where Onureg significantly improved overall survival and relapse-free survival in patients with AML.
It is approved in the United States for continued treatment of adult patients with AML who achieved first CR or CR with incomplete blood count recovery following intensive induction chemotherapy and are not able to complete intensive curative therapy.
The approval of the drug in additional geographies will augment Bristol Myers’ strong oncology portfolio, which consists of drugs like Revlimid, Opdivo, Pomalyst, Sprycel and Yervoy, among others.
Bristol-Myers’ shares have gained 7.8% year to date against the industry's decline of 1.8%.
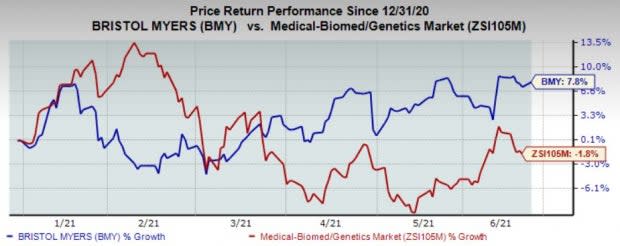
Image Source: Zacks Investment Research
The company’s performance in the first quarter of 2021 was dismal as its immuno-oncology drug Opdivo’s sales declined facing stiff competition from the likes of Merck’s MRK Keytruda. Moreover, Revlimid sales weren’t impressive.
Nevertheless, the recent approval of new drugs adds a stream of revenues, which should propel growth in the coming quarters.
In March, the company and partner bluebird bio, Inc. BLUE secured the FDA approval for Abecma (idecabtagenevicleucel; ide-cel) as the first B-cell maturation antigen (BCMA)-directed chimeric antigen receptor (CAR) T cell immunotherapy for the treatment of adult patients with relapsed or refractory multiple myeloma.
Last month, the FDA gave a nod to Zeposia (ozanimod) 0.92 mg for the treatment of adults with moderately-to-severely active ulcerative colitis (UC), a chronic inflammatory bowel disease (IBD).
Bristol-Myers currently carries a Zacks Rank #3 (Hold). A better-ranked stock in the health care sector is Repligen Corporation RGEN, which presently carries a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Repligen’s earnings estimates for 2021 have increased to $2.26 from $1.91 in the past 60 days.
Infrastructure Stock Boom to Sweep America
A massive push to rebuild the crumbling U.S. infrastructure will soon be underway. It’s bipartisan, urgent, and inevitable. Trillions will be spent. Fortunes will be made.
The only question is “Will you get into the right stocks early when their growth potential is greatest?”
Zacks has released a Special Report to help you do just that, and today it’s free. Discover 7 special companies that look to gain the most from construction and repair to roads, bridges, and buildings, plus cargo hauling and energy transformation on an almost unimaginable scale.
Download FREE: How to Profit from Trillions on Spending for Infrastructure >>
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
Repligen Corporation (RGEN) : Free Stock Analysis Report
bluebird bio, Inc. (BLUE) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

