AstraZeneca withdraws Covid vaccine worldwide after admitting it can cause rare blood clots
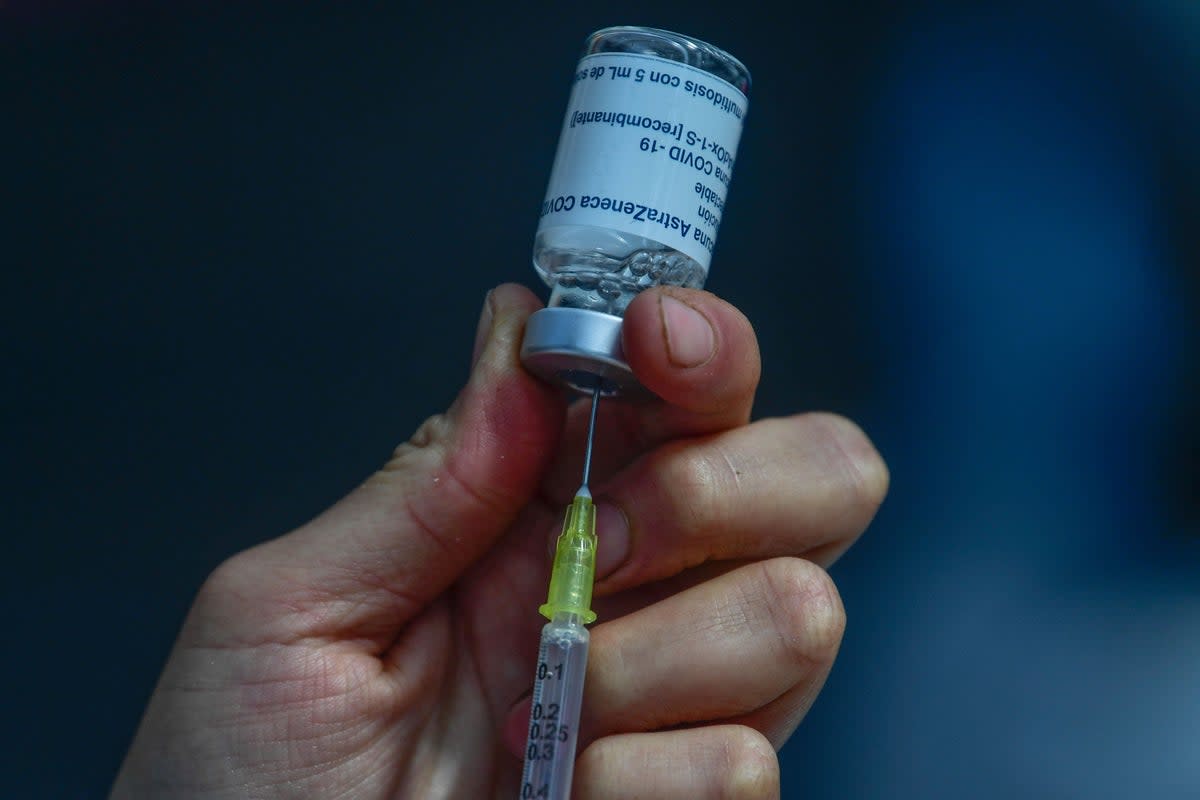
AstraZeneca is withdrawing its Covid-19 vaccine worldwide, months after the pharma giant admitted the drug could cause very rare, but life-threatening, injuries.
The British-Swedish drugmaker has already withdrawn its EU marketing authorisation for the vaccine, branded Vaxzevria since 2021. The authorisation is the approval to market a drug in EU’s member states. The withdrawal was due to a “surplus of available updated vaccines” against new variants of the novel coronavirus, the company said.
The application to withdraw the vaccine from the EU was made on 5 March and came into effect on 7 May.
“As multiple variant Covid-19 vaccines have since been developed there’s a surplus of available updated vaccines," AstraZeneca said, adding that this led to a fall in demand for Vaxzevria, which is no longer manufactured or supplied.
AstraZeneca recently admitted that its vaccine, initially called Covishield, could cause very rare side effects like blood clots and low blood platelet counts, The Telegraph reported.
The admission came after the company was slapped with a class action lawsuit in the UK, which claimed that the vaccine had caused deaths and severe injuries and sought damages up to £100m for about 50 victims.
“It is admitted that the AZ vaccine can, in very rare cases, cause TTS. The causal mechanism is not known,” AstraZeneca said in court documents in February, the newspaper reported.
TTS is thrombosis with thrombocytopenia syndrome, which is characterised by blood clots and low blood platelet counts in humans.
AstraZeneca’s vaccine was developed in collaboration with Oxford University and produced by the Serum Institute of India. It was widely administered in over 150 countries, including Britain and India.
Some studies conducted during the pandemic found the vaccine was 60 to 80 per cent effective in protecting against the novel coronavirus.
But subsequent research found that it caused some people to develop potentially fatal blood clots.
AstraZeneca’s admission that the vaccine could potentially prove lethal ran counter to its insistence in 2023 that it would “not accept that TTS is caused by the vaccine at a generic level”.
In April 2021, the World Health Organisation also confirmed that the vaccine could have fatal side effects. “A very rare adverse event called thrombosis with thrombocytopenia syndrome, involving unusual and severe blood clotting events associated with low platelet counts, has been reported after vaccination with this vaccine.”
In a statement, AstraZeneca said: “We are incredibly proud of the role Vaxzevria played in ending the global pandemic. According to independent estimates, over 6.5 million lives were saved in the first year of use alone, and over three billion doses were supplied globally.
“Our efforts have been recognised by governments around the world and are widely regarded as being a critical component of ending the global pandemic.”


